Cytochrome P450 enzymes

Cytochrome P450 enzymes (P450, CYP) are a superfamily of heme-thiolate proteins. In humans, they are indispensable for detoxification in the liver, steroid hormone biosynthesis, and fatty acid metabolism.
Many P450 enzymes are involved in disease pathogenesis, such as hypertension, cardiac disease, traumatic brain injury, and cancer. Thus, they are exciting drug targets and offer transformative opportunities for new therapy options.
1. Elucidating the Role of P450 omega-hydroxylases in human health and disease
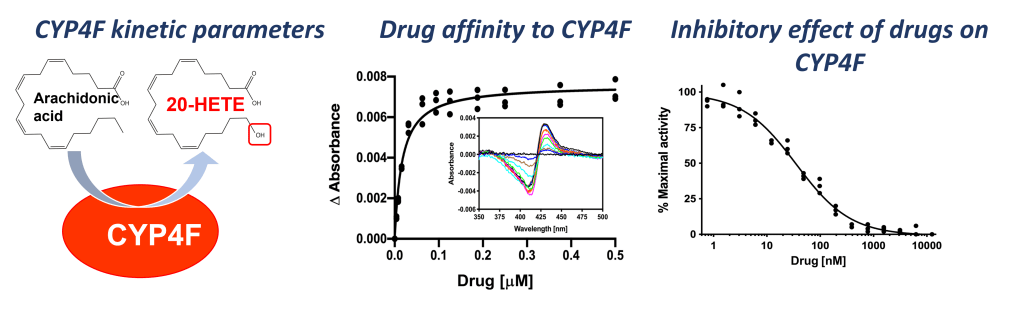
Members of the CYP4F family belong to a group of w-hydroxylases which produce important lipid mediators in the human body. One of these lipid mediators is the molecule 20-hydroxyeicosatetraenoic acid (20-HETE) which is regulating the blood pressure and promotes the formation of new blood vessels in healthy humans. However, 20-HETE has been identified as a culprit in human disease and thus, 20-HETE generating P450 might be exciting new drug targets for cancer treatment.


We use a combination of cell biology and biochemistry to assess the role and function of CYP4F enzymes in physiology and disease. Moreover, we use X-ray protein crystallography to solve structures of CYP4F enzymes for directed design of selective drugs which only inhibit the target and not any of the other 56 P450s in the human body.
2. Structural Characterization of ancestral CYP11A1 enzymes
Human CYP11A1 is crucial for steroid hormone biosynthesis in the human body. It metabolizes cholesterol which is the common precursor for steroid hormones which are divided into glucocorticoids, mineralocorticoids, and sex hormones.
Recently, the common and the mammalian ancestor of CYP11A1 was resurrected to study the evolution of these crucial human enzymes. Interestingly, ancestral CYP11A1 isoforms exhibit a different substrate preference compared to human CYP11A1 which might provide clues for their evolution.
In collaboration with Dr. Rita Bernhardt from Saarland University (Germany), we will use X-ray protein crystallography to examine the structural basis for an altered substrate specificity.
